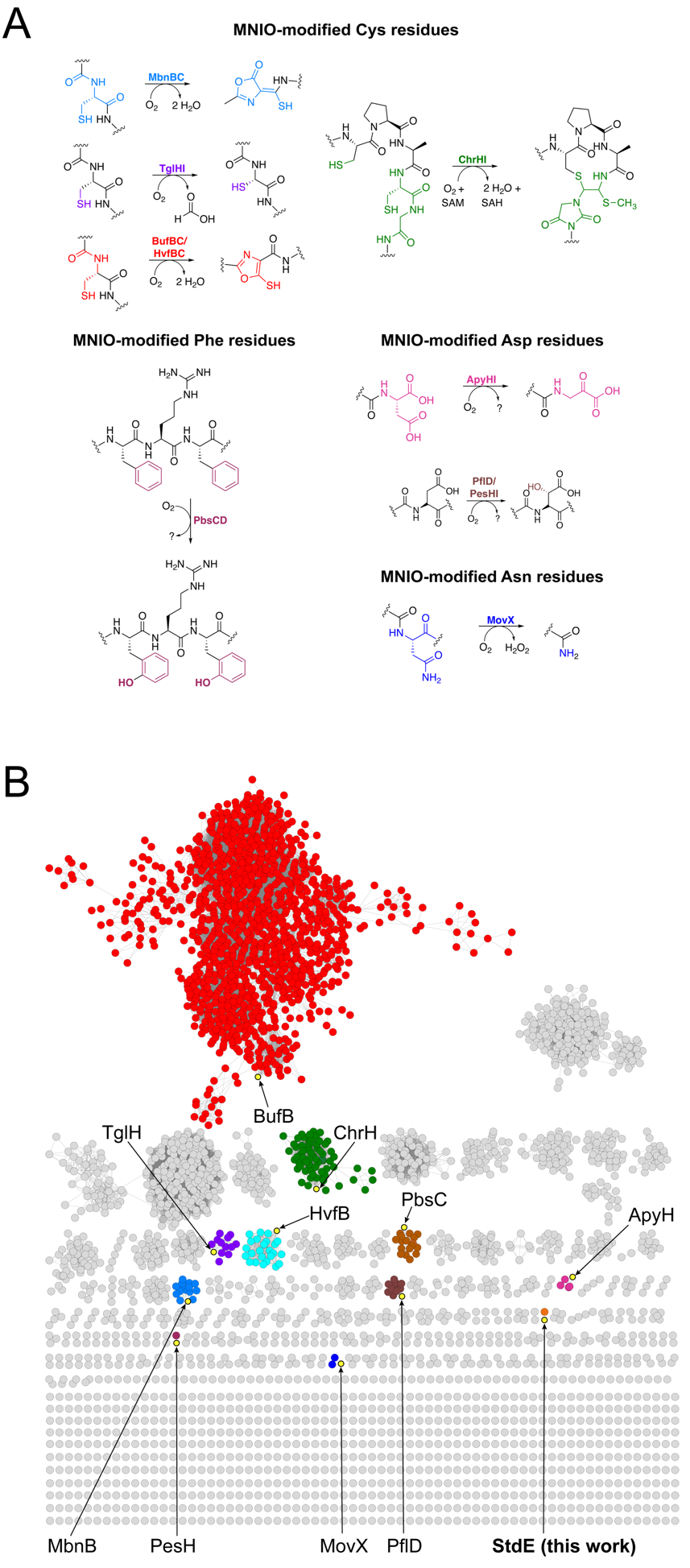
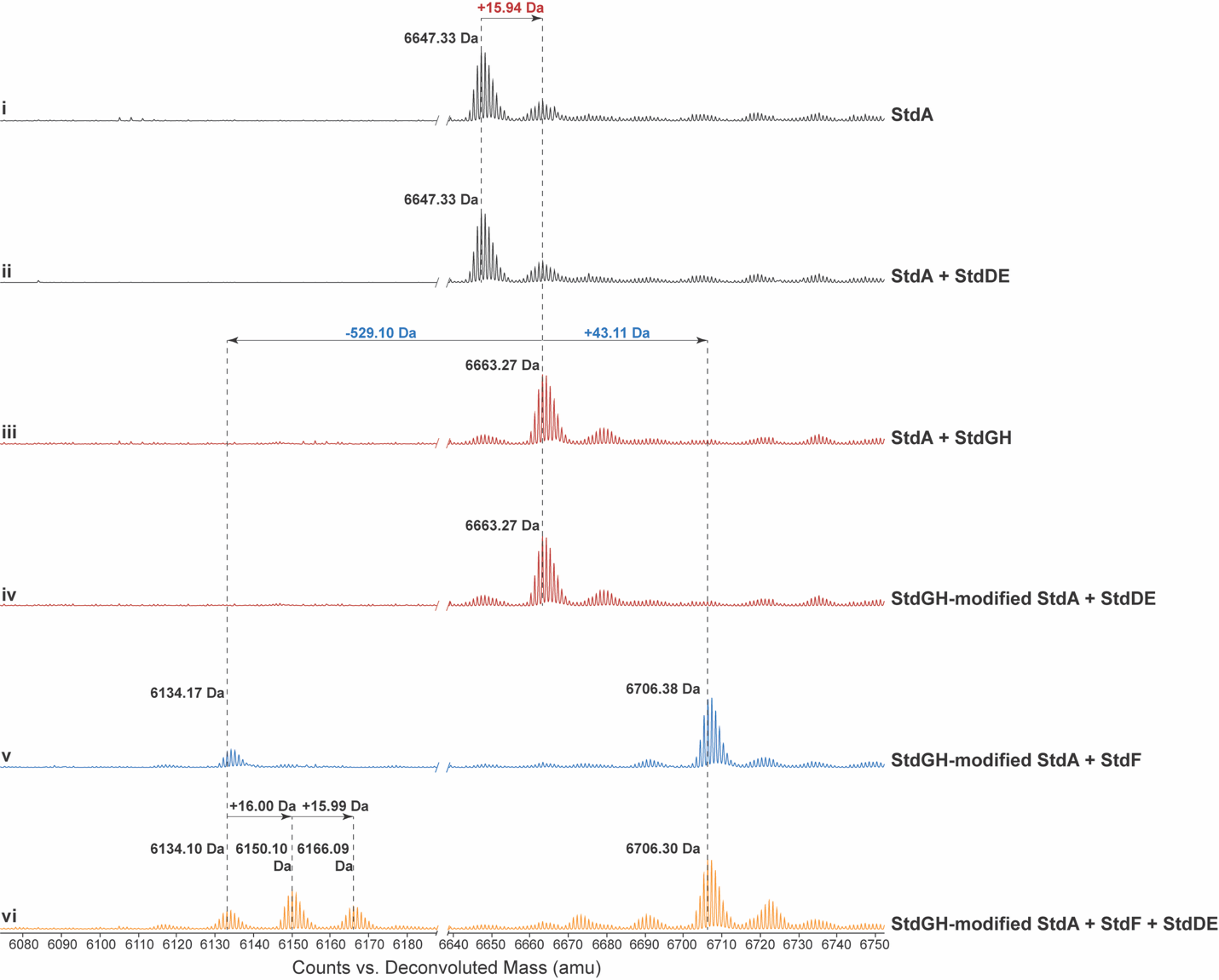
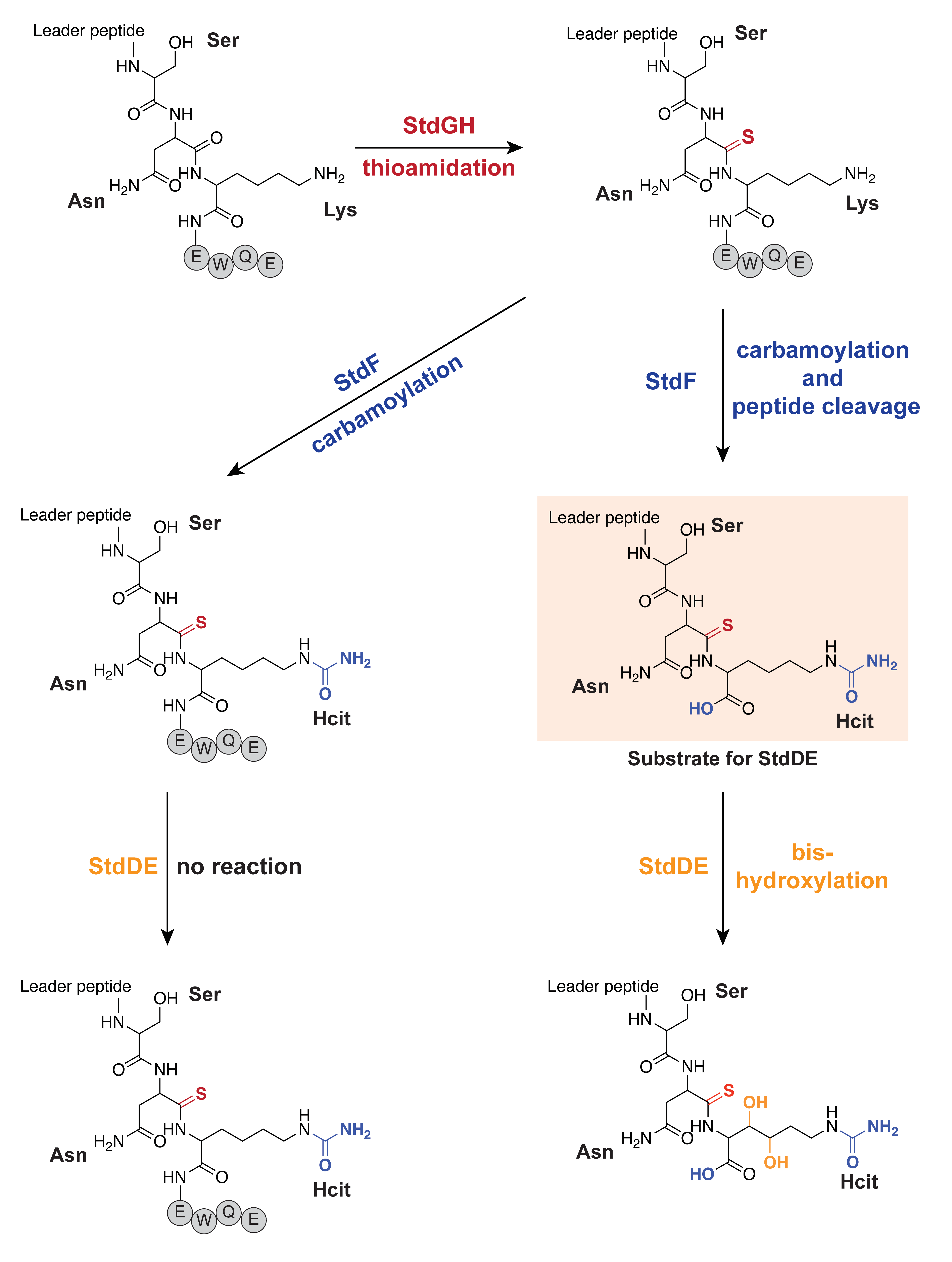
StdGH enzymatic assay. Lyophilized StdA was dissolved in buffer (50 mM HEPES pH 8, 150 mM KCl) to a final concentration of 1 mM and then frozen in individual aliquots at -80 °C until used. For the first step of the biosynthesis pathway (thioamidation), aliquots of StdA and purified StdGH were thawed on ice. The 200 µL reaction mixture contained 100 µM StdA, 20 µM StdGH, 50 mM HEPES pH 8, 150 mM KCl, 5 mM Na2S, 5 mM ATP, 5 mM MgCl2, and 1 mM DTT. The reaction was incubated at room temperature overnight on the bench. The next day, the reaction mixture was heated to 70 °C for 10 min then centrifuged at 8000 × g to remove StdGH. The supernatant containing the StdGH-modified StdA product was dialyzed into Buffer C using a 2K MWCO Slide-A-Lyzer™ Dialysis Cassette, following the manufacturer’s instructions to remove the excess Na2S, ATP, MgCl2, and DTT. The samples were then analyzed directly using intact mass spectrometry described above.
StdF enzymatic assay. StdGH-modified StdA was used as the substrate for the StdF enzyme assay. The 200 µL reaction mixture contained 100 µM StdGH-modified StdA, 20 µM StdF, 50 mM HEPES pH 8, 150 mM KCl, 1 mM carbamoyl phosphate, 5 mM ATP, and 5 mM MgCl2. The reaction was incubated at room temperature overnight on the bench. The following day, the reaction mixture was heated to 70 °C for 10 min and then centrifuged at 8000 × g to remove StdF. The supernatant containing the StdGHF-modified StdA product was then analyzed directly using intact mass spectrometry described above.
StdDE enzymatic assay. The StdDE enzyme assay was performed in the presence of StdF to immediately transfer the cleaved, carbamoylated, and thioamidated substrate to StdDE. StdDE was prepared with a 5-fold molar excess of ascorbate and ammonium iron(II) sulfate hexahydrate on the benchtop before the reaction. The 200 µL reaction mixture contained 100 µM StdGH-modified StdA, 20 µM StdF, 20 µM StdDE, 50 mM HEPES pH 8, 150 mM KCl, 1 mM carbamoyl phosphate, 5 mM ATP, 5 mM MgCl2, 100 µM ascorbate, and 100 µM ammonium iron(II) sulfate hexahydrate. The reaction was incubated on the bench at room temperature overnight. The following day, the reaction mixture was heated to 70 °C for 10 min, then centrifuged at 8000 × g to remove StdF and StdDE. The supernatant containing StdGHFDE-modified StdA was then analyzed using intact mass spectrometry described above.
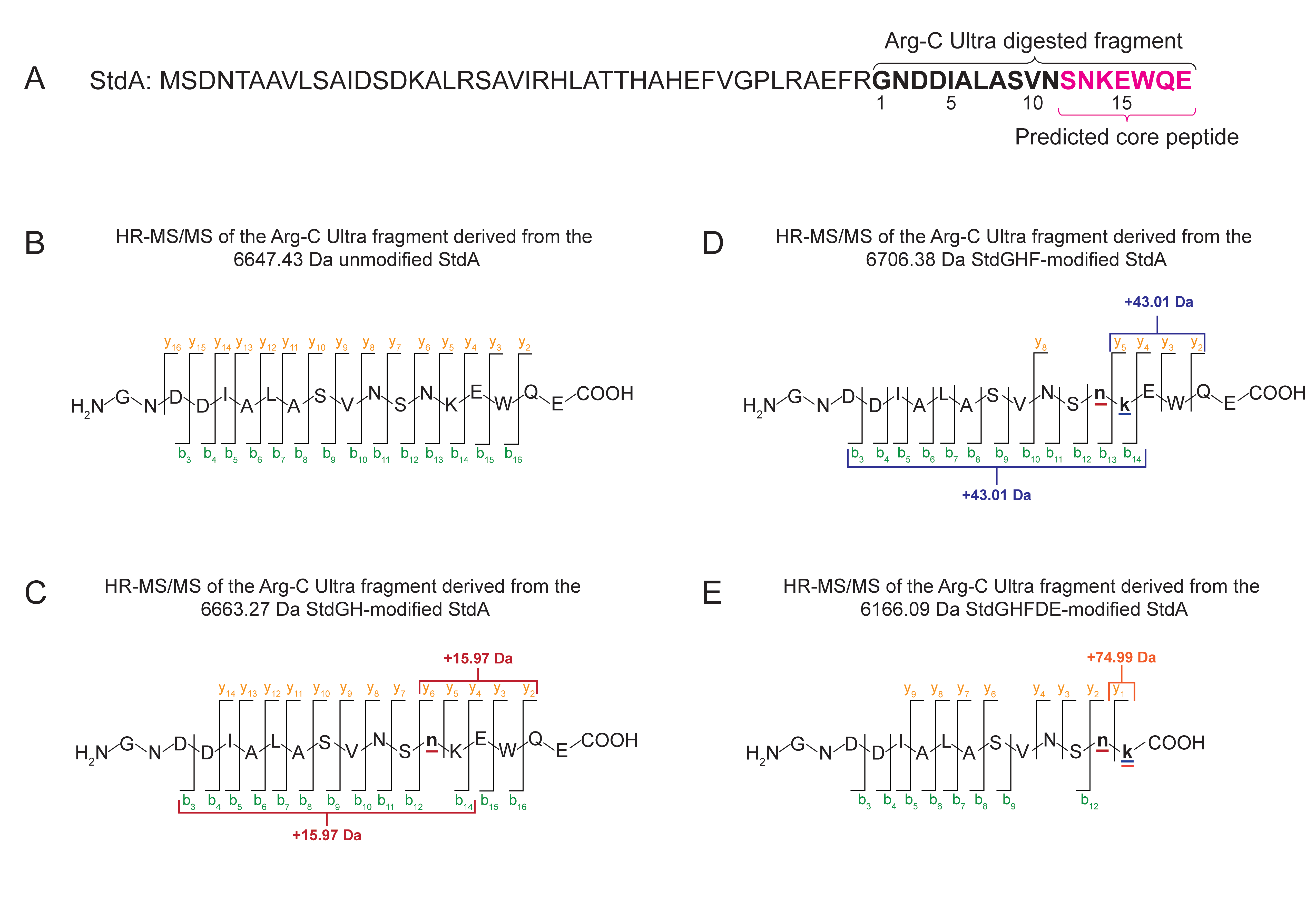
Isolation of unmodified and modified core-containing fragments. To isolate unmodified and modified core-containing fragments, proteolysis was performed for 45 min at 37 °C using ArgC-Ultra at an enzyme-to-substrate ratio of 1:500. The reactions were then quenched by adding formic acid to a final concentration of 0.1%. HPLC purification was performed to isolate the fragments on an Agilent 1100 system in the Northwestern Keck Biophysics Facility. The column used was a Phenomenex Aeris Peptide XB-C18 LC column (Part no. 00G-4632-N0; particle size: 5 µm; dimensions: 250 x 10 mm; pore size: 100 Å) coupled with a SecurityGuard SemiPrep Cartridge core-shell C18-Peptide (Part no. AJ0-9317; dimensions: 10 x 10 mm). The mobile phase consisted of solvent A, which contained water with 0.5% acetic acid, and solvent B, with acetonitrile and 0.5% acetic acid. The column was equilibrated with 10 column volumes of 99% solvent A and 1% solvent B. An injection volume of 500 µL was used. Separation was performed over a gradient of 1-30% solvent B at a flow rate of 2 mL/min for 30 min. The column was then washed with 95% solvent B for 5 min at 2.5 mL/min, followed by re-equilibration with 1% solvent B for 10 min at 2 mL/min. The products were freeze-dried to remove the acetonitrile/water/0.5% acetic acid solution. Retention times of the Arg-C Ultra digested unmodified StdA, StdAGH-modified StdA, StdAGHF-modified StdA, and StdAGHFDE-modified StdA are listed in Table S11.
High-resolution tandem mass spectrometry (HR-MS/MS). HR-MS/MS data collection and analysis were performed in the Northwestern Proteomics Core Facility. Briefly, the raw data for the Arg-C Ultra digested samples were acquired on an Orbitrap Exploris 240 (Lucy) using data-dependent acquisition (DDA) mode. The data were processed in FragPipe against the reference peptide sequence with a universal list of common contaminants included.
Large-scale enzymatic reactions to obtain Arg-C Ultra digested unmodified 15N/13C-Asn13-Lys14 StdA. To obtain the appropriate sample amount for NMR, the unmodified 13C and 15N labeled StdA (20 mg) was dissolved in 5 mL of buffer consisting of 50 mM HEPES pH 8, 150 mM KCl. Arg-C Ultra digestion was performed as described above. The digestion was then sent to the Peptide Synthesis Core Facility of the Center of Regenerative Nanomedicine at Northwestern University for purification of the Arg-C Ultra digested unmodified 15N/13C-Asn13-Lys14 StdA.
Large-scale enzymatic reactions and purification to obtain Arg-C Ultra digested StdGHFDE-modified 15N/13C-Asn13-Lys14 StdA. To obtain the appropriate sample amount for NMR, the unmodified 13C and 15N labeled StdA (65 mg) was dissolved in 10 mL of buffer consisting of 50 mM HEPES pH 8, 150 mM KCl, 20 mM ATP, 20 mM MgCl2, and 20 mM Na2S. The StdGH enzyme complex was then added to the mixture at a final concentration of 100 μM. The reaction was conducted overnight on the benchtop at room temperature. The next day, the reaction was aliquoted into 1.5 mL microcentrifuge tubes and heated at 70 °C for 10 min to remove StdGH. The remaining StdGH-modified 15N/13C-Asn13-Lys14 StdA was dialyzed into Buffer C using a 2K MWCO Slide-A-Lyzer™ Dialysis Cassette, following the manufacturer’s instructions to remove the excess Na2S, ATP, MgCl2, and DTT. The 20 mL reaction mixture for the combined StdF and StdDE reaction contained 500 μM StdGH-modified 15N/13C-Asn13-Lys14 StdA, 100 μM StdF, 100 μM StdDE, 50 mM HEPES pH 8, 150 mM KCl, 1 mM carbamoyl phosphate, 5 mM ATP, 5 mM MgCl2, 500 µM ascorbate, and 500 µM ammonium iron(II) sulfate hexahydrate. The reaction was left on the benchtop at room temperature overnight. The next day, aliquots were heated at 70 °C to remove the enzymes and centrifuged at 8000 × g to obtain the supernatant. Arg-C Ultra digestion was performed as described above. The digestion was then sent to the Peptide Synthesis Core Facility of the Center of Regenerative Nanomedicine at Northwestern University for purification of the Arg-C Ultra digested StdGHFDE-modified 15N/13C-Asn13-Lys14 StdA.
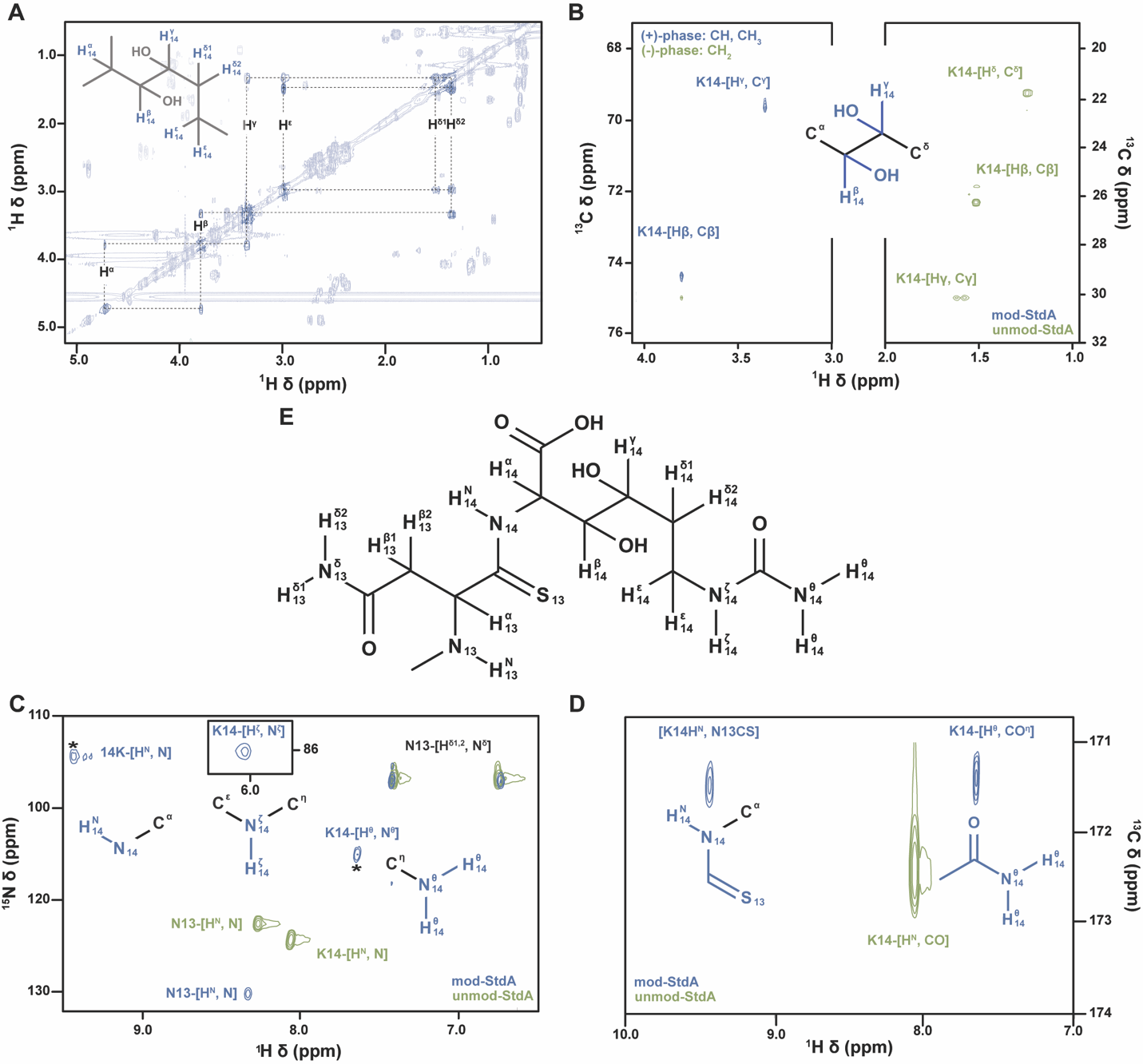
NMR data acquisition and analysis. All spectra were collected at 25 °C on a Bruker HCFN 600 MHz spectrometer at Northwestern’s IMSERC facility, equipped with a triple channel, QCI cryo probe with Z gradients. Assignment experiments were carried out at natural 13C/15N abundance on the modified StdA peptide at a concentration of 200 µM in both H2O and D2O using uniform spectral sampling. The TOCSY experiments were collected using States-TPPI acquisition with sweep widths of 8197 and 7198 Hz in the F2 and F1 dimensions with respective resolutions of 4.0 and 28.1 Hz in both solvents. Homonuclear Hartman-Hahn transfers were accomplished with DIPSI2 and excitation sculpting was achieved with gradients. ROESY experiments were acquired in States-TPPI mode with sweep widths of 7143 Hz in both F2 and F1 and resolutions of 3.5 and 27.9 Hz in F2 and F1 in both solvents. A cw spinlock was used with a mixing time of 200 ms. A D2O-COSY was collected using QF acquisition with sweep widths of 7143 and 7198 Hz in F2 and F1 with respective resolutions of 3.5 and 28.1 Hz. Gradient pulses were used for selection. A multiplicity-edited, D2O 13C-HSQC was collected using echo-antiecho acquisition with sweep widths of 8197 and 24887 Hz in F2 and F1 with respective resolutions of 8.0 and 388.9 Hz. Magnetization transfer was accomplished using double INPET transfers optimized for sensitivity enhancement and decoupling was used during acquisition.
The HNCO, low pH 15N-HSQCs, and comparative 13C-HSQCs were collected on unmodified and StdGHFDE-modified U-[13C,15N]-Asn13-Lys14-StdA at 1500 and 700 μM, respectively, in 20 mM sodium phosphate pH 6.8, 50 mM NaCl, 1% DSS, 10% D2O. The 15N-HSQCs were non-uniformly sampled, phase-sensitive, double INPET transfer-based sequences with Watergate solvent decoupling. They had 15N offsets of 105 ppm with sweep widths of 6849 and 3039 Hz in 1H and 15N, leading to resolutions of 6.7 and 23.7 Hz respectively. The HNCOs were acquired as pseudo-2D experiments with one time domain point for the 15N dimension, sweep widths of 8197 and 4525 Hz in 1H and 13C giving resolutions of 8.0 and 70.7 Hz respectively. The 13C-HSQCs were non-uniformly sampled, multiplicity-edited, echo-antiecho acquired, double INPET transfer based sequences. A 13C offset of 40 ppm was used with sweep widths of 8197 and 12066 Hz in F2 and F1 with respective resolutions of 8 and 94.3 Hz.
ASSOCIATED CONTENT
Supporting Information
Supplementary Tables S1-S11, Supplementary Figures S1-S14