Using Patient iPSC-derived Retinal Pigment Epithelial Cells to Evaluate Differential Susceptibility to MEK Inhibitor-Associated Retinopathy
Lola P. Lozano¹,² , Timothy M. Boyce¹,², Andrew P. Groves⁴, Henry L. Keen⁵, Culver Boldt¹,², Robert F. Mullins¹,², Elaine M. Binkley¹,², Budd A. Tucker*¹,²,³
¹Institute for Vision Research, University of Iowa, Iowa City, IA, 52242, USA. ²Department of Ophthalmology and Visual Sciences, Carver College of Medicine, University of Iowa, Iowa City, IA, 52242, USA. ³Department of Neuroscience and Pharmacology, Carver College of Medicine, University of Iowa, Iowa City, IA, 52242, USA. ⁴Stead Family Department of Pediatrics, The University of Iowa, Iowa City, IA, 52242, USA. ⁵Iowa Institute of Human Genetics, Carver College of Medicine, University of Iowa, Iowa City, IA, 52242, USA.
Abstract
Purpose: Compare the effect of MEK inhibition on iPSC-derived retinal pigmental epithelial (RPE) cells generated from a patient who developed MEK inhibitor-Associated Retinopathy (MEKAR) versus a patient who did not develop retinopathy.
Design: Case-control
Subjects: Two female patients with Neurofibromatosis Type 1 who were treated with MEK inhibitors. One patient developed MEKAR, the other did not.
Methods: RPE were generated from human induced pluripotent stem cells (hiPSCs) from these two patients. These hiPSC-derived RPE were treated with selumetinib for 10 days.
Main Outcome Measures: Phagocytic activity and changes in gene expression
Results: As previously reported, there was a significant increase in internalized rhodopsin in phagocytosis assays, yet this was only found in hiPSC-derived RPE from the patient who developed MEKAR. Selumetinib decreased expression of genes related to fluid transport and cell volume, including aquaporins and solute transporters. At baseline, cells from the patients without MEKAR had higher expression of these genes. Interestingly, selumetinib-induced changes in gene expression only reached statistical significance in cells from the patient who did not develop MEKAR, suggesting these changes may be a compensatory protective mechanism. Patients susceptible to forming MEKAR may have increased phagocytosis without a compensatory change in expression of genes related to fluid flux, thereby inhibiting their ability to transport fluid out of the subretinal space.
Conclusions: MEK inhibitor-Associated Retinopathy may only affect susceptible patients whose retinal pigment epithelium cannot sufficiently regulate expression of genes related to fluid transport and cell volume, altering the ability of these cells to properly function.
Keywords: Retinopathy, MEK Inhibitor-Associated Retinopathy, retinal pigment epithelium, induced pluripotent stem cells, ocular oncology, eye tumors, neurofibromas, chemotherapy side effects
Introduction
MEK inhibitors are widely used in oncology to treat many types of malignancies. In particular, these drugs are used long term in patients with Neurofibromatosis Type 1 (NF1) related neurofibromas. Due the ubiquity of the MEK pathway in cell signaling, many patients experience side effects. One side effect that can significantly decrease patients’ quality of life and lead to treatment cessation is MEK inhibitor-associated retinopathy (MEKAR). MEKAR is characterized by subretinal fluid accumulation that leads to decreased visual acuity.
While the mechanism of MEKAR in unknown, it is hypothesized to cause dysfunction of the retinal pigment epithelium (RPE), preventing these cells from transporting fluid from the subretinal space to the choroid. In a previous study we demonstrated that inhibition of MEK (via treatment with 10 μM selumetinib) in mature human iPSC-derived RPE cells led to increased phagocytosis and differential expression of genes related to fluid transport and cell volume regulation. Furthermore, these findings were detectable by as little as 10 days of treatment.
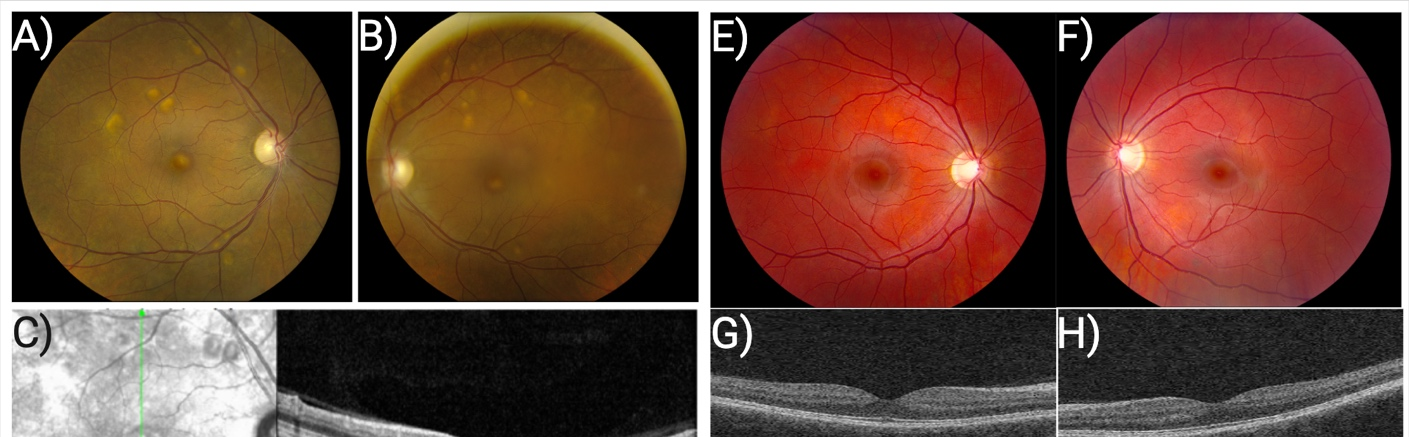
In this study, we compared hiPSC-derived RPE generated from two female patients with NF1 who were both treated systemically with MEK inhibitors yet only one of them developed MEKAR (Table 1, Figure 1). To determine if MEK inhibition has a differential effect on hiPSC-derived RPE cells between these patients, we treated cells for ten days and measured changes in phagocytosis and expression of genes relevant to fluid transport.
Methods
Study Approval
This study received approval from the University of Iowa Institutional Review Board (IRB No. 200202022). Both patients provided informed consent, and research was conducted in accordance with the Declaration of Helsinki.
Genetic Sequencing
DNA from patient-derived fibroblasts was isolated and submitted for sequencing at the Iowa Institute of Human Genetics – Genomics Division. High–molecular weight genomic DNA was extracted and quantified using the Qubit dsDNA HS Assay. For library preparation, 3–5 µg of genomic DNA was sheared to a target size of ~15 kb using Covaris g-Tubes by centrifugation at 4,700 × g for 60 s, followed by tube inversion and a second centrifugation under identical conditions. Sequencing libraries were prepared using the Oxford Nanopore Technologies (ONT) Ligation Sequencing Kit following the manufacturer’s instructions.
Sequencing was performed on PromethION R10.4.1 flow cells. Adaptive sampling was configured to target chromosome 17. High-accuracy base-calling with methylation detection (5mC in CpG context) was performed and unaligned bam/fastq files were generated. Nextflow was used for running all bioinformatics workflows. Small variants were identified using Clair3 and large structural variants using Sniffles2. VCF files were filtered for read depth (DP > 10) and genotype quality (GQ > 10). Annotation was performed using the Ensembl Variant Effect Predictor. Phase analysis was performed using WhatsHap.
Generation of Human Induced Pluripotent Stem Cells
Human induced pluripotent stem cells (hiPSCs) were generated from 2 female patients with NF1. Fibroblasts were isolated and expanded from a 3 mm dermal biopsy and transduced with CytoTune-iPSC 2.0 Sendai Reprogramming Kit. hiPSC colonies were isolated and expanded. TaqMan Human Pluripotent Stem Cell Scorecard Panel was used to analyze pluripotency and loss of viral transgene expression. Karyotype analysis was carried out following a standard G-banding protocol. Cells were maintained at 37°C, 5% CO₂, and 20% O₂.
Differentiation of hiPSC-derived Retinal Pigment Epithelial Cells
A rapid, directed differentiation protocol was followed. The ROCK-inhibitor Y-27632 was added to cell media at each passage for 4-7 days to increase cell survival and attachment. hiPSCs were grown to 80% confluence in Essential 8 Flex media on rhLaminin-521 coated plates and then passaged onto Matrigel-coated plates and treated for 14 days with growth factors. After RPE expansion, cells were maintained in RPE Supporting Media (RSM) on Matrigel-coated plates. Cells were passaged after reaching confluence every 4-5 days with TypLE Express and plated at a seeding density of 2.5×10⁴ cells/cm². Only cells at or below passage 3 were used. Cells were characterized for cell identity using gene and protein expression. Once cells were seeded onto experimental plates at density of 1.0×10⁵ cells/cm² they were allowed to mature for 30 days before drug treatment.
Pharmacologic Inhibitor
Selumetinib (MEK inhibitor) was reconstituted to 10 mM in DMSO per the manufacturer’s recommendations. Confirmation of drug efficacy at 10 μM was assessed via Western Blot for ratio of Ph-MAPK to total MAPK.
Gene Expression Analysis
Cells were washed with 1× DPBS then incubated with TrypLE for 5 minutes at 37°C. cDNA was generated from RNA using SuperScript IV VILO Master Mix. PCR reactions were run using PrimeTime qPCR probes and TaqMan Fast Advanced Master Mix. Data were analyzed using delta-delta Ct method normalized to the B2M housekeeping gene.
Rod Outer Segment Phagocytosis Assay
Bovine rod outer segments (bROS) at a concentration of 10 bROS/cell were added to hiPSC-derived RPE cells. Cells were incubated with bROS for 3 hours at 37°C and 5% CO₂. After 3 hours, wells were washed five times with 1× DPBS with calcium and magnesium. For the Internalized bROS condition, wells were incubated with 2 mM EDTA in PBS for 10 minutes at 37°C, 5% CO₂ to remove unbound bROS.
Western Blot Analysis
Cells were lysed with RIPA Lysis and Extraction Buffer with 1:100 Halt Phosphatase and Proteinase Cocktail Inhibitors. Protein concentration was quantified using Pierce BCA Protein Assay. 5 µg protein was run on a 4-20% Novex Tris-Glycine gel for 25 minutes at 225 volts. Protein transfer was performed using iBlot 3 at 25 volts. Blots were probed with primary antibody then HRP-conjugated secondary antibody and imaged using iBright FL1500. Densitometry was performed in FIJI ImageJ with all protein bands normalized to beta-actin.
Statistical Analysis
Statistical analysis was performed in GraphPad Prism Version 10.2.3. All data were collected from three independent experiments (n=3). Statistical significance was calculated using paired t-test for all RPE cell function assays (alpha = 0.1).
Results
Identification of Pathogenic Mutations
The NF1 patient with MEKAR had a premature STOP mutation (C>T) at chr17.31229061 and the NF1 patient without MEKAR had a frameshift mutation (G>GT) at chr17.31233203.
Impact of MEK Inhibition on Rod Outer Segment Phagocytosis
There was no difference in total RHO between untreated and treated conditions for each patient (1.9 vs 2.2 for patient with MEKAR, 1.7 vs 1.6 for patient without MEKAR). There was, however, a significant increase in internalized RHO in treated cells from the patient with MEKAR (1.2 vs 1.9, p<0.05) and no significant difference in the patient without MEKAR (1.4 vs 1.5).
Differential Gene Expression Between MEKAR and Non-MEKAR iPSC-derived RPE Cells
Selumetinib decreased expression of AQP1, AQP11, ATP6V1C2, LRRC8C, SLC6A6, SLC12A2, SLC16A1, and SLC22A23 in both patient lines. Expression of AQP3 and AQP7 were unaffected by MEK inhibition. When iPSC-derived RPE generated from the MEKAR patient were compared to the non-MEKAR patient, with the exception of AQP11, all genes evaluated were expressed at higher levels in non-MEKAR iPSC-derived RPE. Differences in expression were found to be statistically significant for the genes AQP1, AQP3, AQP7, and CLCN5.

Discussion
The present study contributes to our understanding of the pathophysiology underlying MEKAR by testing a mechanistic hypothesis directly in a human, patient-derived RPE system: that susceptibility to MEKAR reflects differential vulnerability of the retinal pigment epithelium to MEK inhibition. This is clinically and anatomically plausible because the RPE is the key epithelial layer responsible for maintaining outer retinal adhesion and preventing subretinal fluid accumulation via tightly regulated ion and water transport.
The most direct functional divergence observed between donor lines was seen in the phagocytosis assay: specifically, while selumetinib exposure produced a statistically significant increase in internalized rhodopsin in iPSC-derived RPE cells derived from the MEKAR patient (1.9 vs 1.2), iPSC-derived RPE from the non-MEKAR patient showed no significant change (1.5 vs 1.4). Internalized rhodopsin is metabolized by RPE to ketones which are recycled back into the subretinal space. The polarity of these anions attract sodium and water. Thus, MEK inhibitor induced increases in internalization of outer segments in vivo may lead to subretinal fluid accumulation and the MEKAR phenotype.
The present study also found that selumetinib recapitulates directional changes in a focused panel of genes implicated in fluid transport and cell volume regulation, but the pattern of statistical significance differs markedly between lines: the non-MEKAR line shows significant decreases across multiple genes, whereas the MEKAR line shows limited significant change (AQP11 only). These findings may suggest that the non-MEKAR line can mount a regulated transcriptional response to MEK inhibition, whereas the MEKAR line is comparatively “transcriptionally inflexible” and instead expresses vulnerability through dysregulated cellular handling of photoreceptor outer segment material.
NF1 is a rare inherited cancer predisposition syndrome, making it challenging to obtain tissue samples for research. We are fortunate to have received biopsies from two patients; however, our sample size is limited to a one-to-one comparison. Additionally, while hiPSC-derived RPE enable in vitro experiments dissecting the effect of MEK inhibitors on a single subtype important for retinal health, the isolated nature of this experimental design prevents conclusions about cell-cell interaction and the broader biological environment that likely play a role in MEKAR pathophysiology. Future work using microphysiologic systems and in vivo work is needed to fully understand the mechanism underlying this retinopathy.
Many oncology patients treated with MEK inhibitors suffer vision loss due to MEK inhibitor-associated retinopathy (MEKAR). Patients who develop MEKAR may have disrupted function of the retinal pigment epithelium (RPE). In response to treatment with the clinically used MEK inhibitor, selumetinib, human induced pluripotent stem cell-derived RPE cells from a patient with MEKAR had increased phagocytosis and decreased expression of genes related to fluid transport compared to a patient who did not develop MEKAR with treatment. Based on these results, we hypothesize that patients who develop MEKAR have an impaired ability to dynamically regulate expression of genes involved in fluid transport when MEK signaling is suppressed—leading to transient failure of outer retina–RPE fluid homeostasis and resultant subretinal fluid. Alternatively, patients who do not develop MEKAR may simply express levels of fluid transporters such as aquaporins and solute carriers that are sufficient to prevent subretinal fluid accumulation despite MEK inhibition induced downregulation. Future work is needed to confirm these findings in vivo and develop appropriate therapies.
Tables
| NF1 with MEKAR | NF1 without MEKAR | |
|---|---|---|
| Sex | Female | Female |
| Age at biopsy (years) | 49 | 21 |
| NF1 Genotype | C to T causing premature STOP at chr17.31229061 | G to GT insertion causing frameshift at chr17.31233203 |
| MEK Inhibitor (dose, frequency, duration) | Selumetinib 50 mg, twice daily 5 months at time of biopsy | Trametinib 2 mg, once daily Treated for 3 months then discontinued prior to biopsy |
Table 1: Sample demographics. Samples were derived from two female patients with NF1 and treated with the same dosage of MEK inhibitors. However, one patient developed MEKAR and the other did not.
References
Ram T, Singh AK, Kumar A, et al. MEK inhibitors in cancer treatment: structural insights, regulation, recent advances and future perspectives. RSC Med Chem . 2023;14(10):1837-1857.
Weber ML, Liang MC, Flaherty KT, Heier JS. Subretinal Fluid Associated With MEK Inhibitor Use in the Treatment of Systemic Cancer. JAMA Ophthalmol . 2016;134(8):855-862.
Jeng-Miller KW, Miller MA, Heier JS. Ocular Effects of MEK Inhibitor Therapy: Literature Review, Clinical Presentation, and Best Practices for Mitigation. Oncologist . 2024;29(5):e616-e621.
Urner-Bloch U, Urner M, Stieger P, et al. Transient MEK inhibitor-associated retinopathy in metastatic melanoma. Annals of Oncology . 2014;25(7):1437-1441.
McCannel TA, Chmielowski B, Finn RS, et al. Bilateral Subfoveal Neurosensory Retinal Detachment Associated With MEK Inhibitor Use for Metastatic Cancer. JAMA Ophthalmology . 2014;132(8):1005-1009.
Francis JH, Habib LA, Abramson DH, et al. Clinical and Morphologic Characteristics of MEK Inhibitor-Associated Retinopathy: Differences from Central Serous Chorioretinopathy. Ophthalmology . 2017;124(12):1788-1798.
Lozano LP, Jennisch M, Jensen R, et al. Modeling MEK inhibitor-Associated Retinopathy in vitro using human induced pluripotent stem cell-derived retinal pigment epithelial cells. bioRxiv . 2025.
Wiley LA, Burnight ER, DeLuca AP, et al. cGMP production of patient-specific iPSCs and photoreceptor precursor cells to treat retinal degenerative blindness. Scientific Reports . 2016;6(1):30742.
Foltz LP, Clegg DO. Rapid, Directed Differentiation of Retinal Pigment Epithelial Cells from Human Embryonic or Induced Pluripotent Stem Cells. J Vis Exp . 2017(128).
Croze RH, Buchholz DE, Radeke MJ, et al. ROCK Inhibition Extends Passage of Pluripotent Stem Cell-Derived Retinal Pigmented Epithelium. Stem Cells Translational Medicine . 2014;3(9):1066-1078.
Yang S, Zhou J, Li D. Functions and Diseases of the Retinal Pigment Epithelium. Front Pharmacol . 2021;12:727870.
Lakkaraju A, Umapathy A, Tan LX, et al. The cell biology of the retinal pigment epithelium. Progress in Retinal and Eye Research . 2020;78:100846.
Lehmann GL, Benedicto I, Philp NJ, Rodriguez-Boulan E. Plasma membrane protein polarity and trafficking in RPE cells: past, present and future. Exp Eye Res . 2014;126:5-15.
Brutsaert EF. Diabetic Ketoacidosis (DKA). MSD Manual . https://www.msdmanuals.com/professional/endocrine-and-metabolic-disorders/diabetes-mellitus-and-disorders-of-carbohydrate-metabolism/diabetic-ketoacidosis-dka . Published 2023. Updated 2023/10.